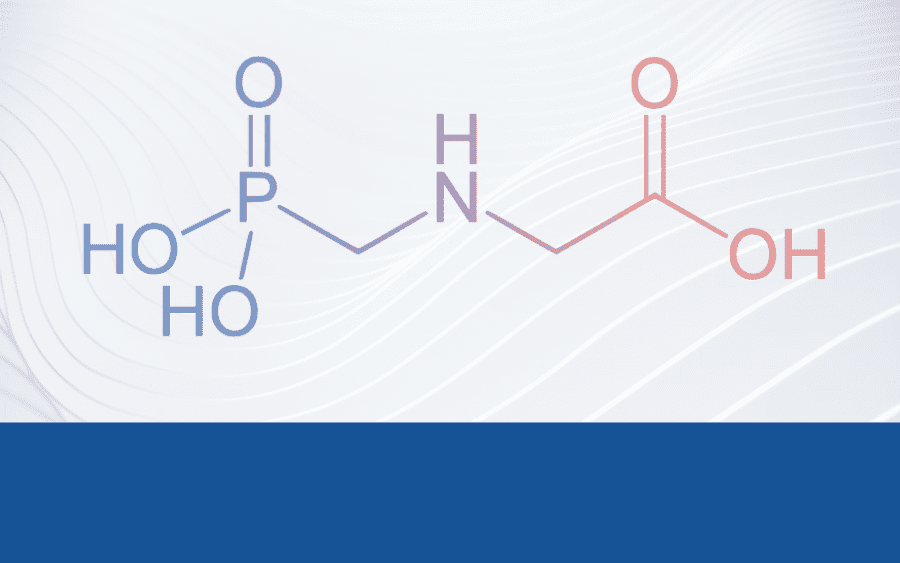
The “Implementing Regulation to renew the approval of glyphosate” was formally adopted on 28 November 2024, as earlier in October, there was no qualified majority either in favour or against in the Committee, in which case, legislation states that the Commission must oblige. The lack of qualified majority is one of the many signs illustrating the long lasting controversy surrounding the substance. Policymakers and experts opposing the legislation cite the numerous gaps in the research related to glyphosate, as well as the range of potential risks connected to it, such as its alleged genotoxic carcinogenic or endocrine disrupting properties. However, the scientific consensus of the European Food Safety Authority (EFSA), based on the dossier prepared by European Chemicals Agency (ECHA) “did not identify critical areas of concern“.
The Commission also acknowledged the potential risks, and included a number of restrictions into the renewal, such as maximum application rates, an “obligation for the applicant to submit information on the possible indirect impacts on biodiversity” and it will prohibit the substance’s use as a desiccant, amongst other rules. In addition, the commission expressed its willingness to review the approval in light of new scientific evidence stemming from the increasing intensity of glyphosate research.
Earlier in 2023 the debate on glyphosate renewal flared up in the parliament, during a dialogue between European Parliament’s Committee on Environment, Public Health and Food Safety (ENVI) and EFSA, preceding the approval’s renewal. EuChemS, following it’s key aim of “providing an unbiased European voice on key policy issues in chemistry and related fields” held a high Impact workshop in the European Parliament on glyphosate in 2017, which concluded that more scientific evidence is needed to properly assess the substance.